Safety Needle Quality Inspection Using AI
The Case
Quality Control for Safety-Engineered Needles
Safety-engineered needles and syringes consist of multiple precision components that must be assembled with high accuracy to ensure safe and reliable use. Even minor deviations can affect needle deployment, reduce functionality, or introduce patient risk during clinical use.
Medical device manufacturers require consistent in-line inspection to verify assembly integrity, reduce defect escape rates, and maintain stable product quality in high-volume production.
Effective safety needle inspection is essential for ensuring product reliability and consistent manufacturing quality.
The Challenge
Detecting Assembly Defects in Low-Contrast Safety-Engineered Needles
Safety needle and syringe assemblies often include small, transparent, reflective, or low-contrast components that are difficult to inspect using traditional inspection systems.
Defects such as protruding needles, misalignment, or incomplete assembly may not be reliably detected using rule-based inspection, particularly under variations in material, surface condition, and lighting.
These limitations reduce inspection robustness, increasing the risk of defective products passing quality control. In high-throughput environments, maintaining consistent detection performance and traceability is difficult with traditional approaches.
The Solution
AI-Based Safety Needle Inspection with SolVision
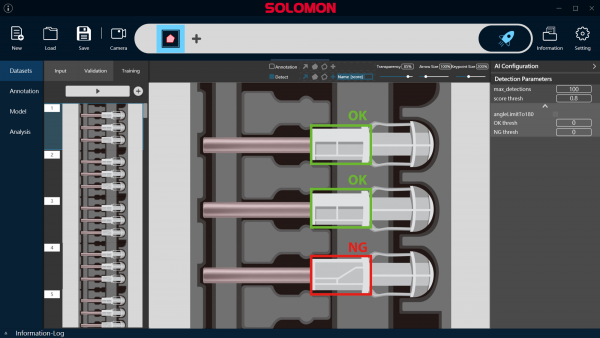
SolVision is an AI vision system software for automated visual inspection of safety-engineered needles in regulated manufacturing environments, supporting QA requirements.
The system is trained on conforming and non-conforming samples to align inspection behavior with QA criteria and device specifications.
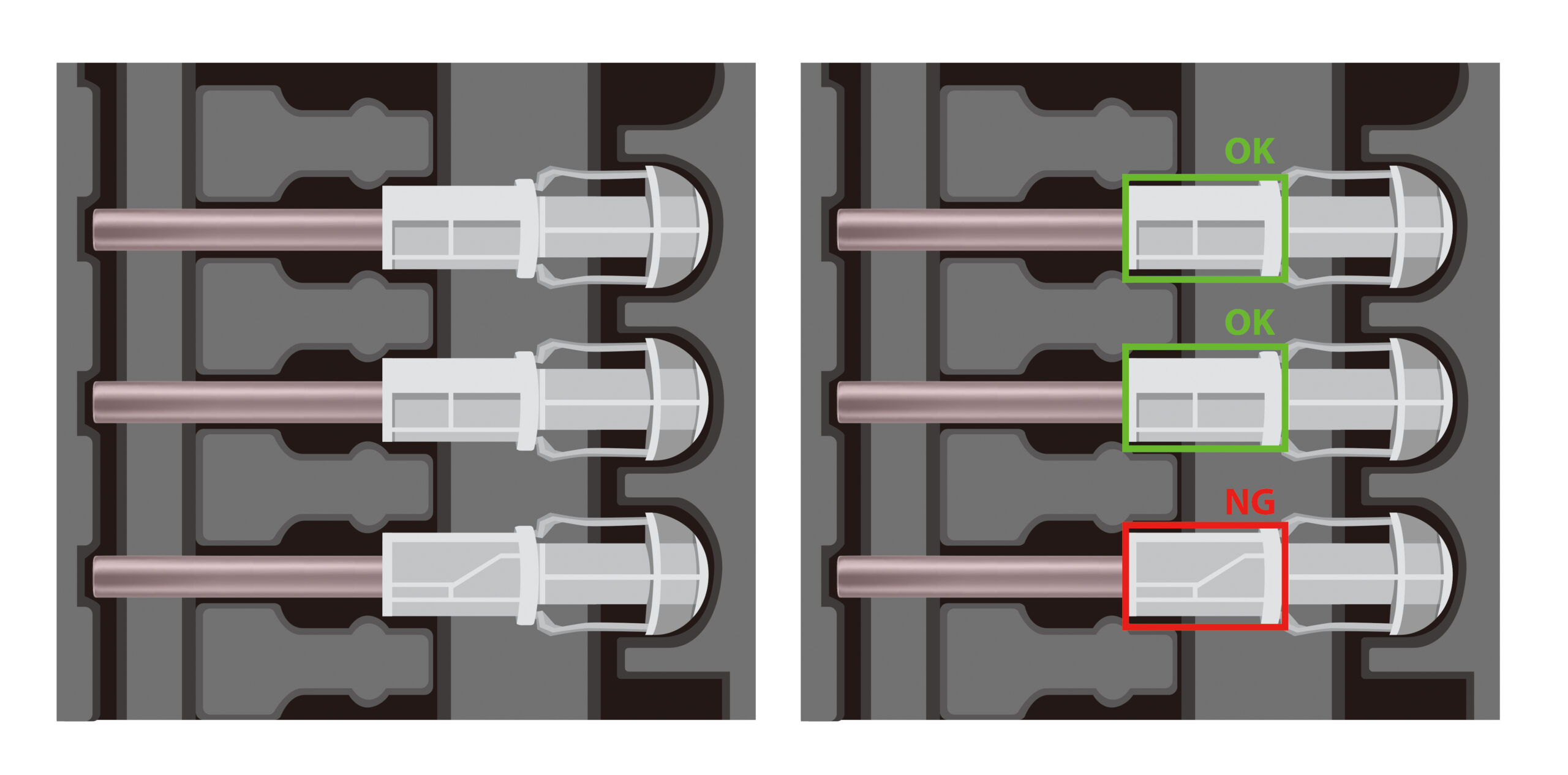
Each unit is inspected to detect assembly deviations, including misalignment, missing components, and incorrect assembly conditions.
Performance remains stable under production variation such as lighting changes, positional shift, and surface variation.
Detected non-conformities include:
- Misaligned or protruding needles
- Missing or incomplete components
- Incorrect assembly conditions
- Defects affecting safety or compliance
Inspection results are recorded for QA documentation, traceability, and batch-level production control.
SolVision enables consistent quality inspection aligned with QA and regulatory requirements for safety-engineered needle production.
SolVision Safety Needle Inspection